Hygiene is a critical concept in food, beverage and life science production. Equipment and facilities, as well as the integrated instrumentation, must be designed to maintain high levels of hygiene and cleanliness during the manufacturing process. The goal is to prevent contamination and ensure that the final product is safe for consumption.
There are several key factors that are typically included in hygienic design. First and foremost, all equipment and surfaces must be designed in such a way that they can be cleaned before each use. In dairies, this is usually done by CIP (Cleaning In Place) systems. To fulfil this requirement, sensors and other equipment must be installed in a self-draining position and without dead spaces. Dead spaces may be found in process connections of sensors, valves, pumps, and other equipment components, but also where piping is connected, in corners and other hard-to-reach areas within processing equipment.
Another important principle of hygienic design is the use of high-quality materials that are free of cracks or crevices where bacteria or other microorganisms can thrive. They also must resist to corrosion and multiple cleaning cycles with aggressive cleaning agents like acids and lye. By ensuring that all equipment used is made of materials like high-quality stainless steel 1.4404 / AISI 316L with a surface roughness equal or better than Ra ≤ 0.8 μm, or of FDA-approved PEEK, processors can ensure hygienic design and cleanability also over long time periods.
Institutions such as 3-A SSI (Sanitary Standards, Inc., a not-for-profit corporation) or the EHEDG (European Hygienic Engineering and Design Group) issue corresponding guidelines according to which engineers have to design the plants, and to which sensor manufacturers can certify their products. Anderson-Negele sensors are developed exclusively for use in hygienic applications. In terms of materials and product design, they are therefore engineered specifically to meet these requirements, and have the corresponding certificates. Nevertheless, when installing the sensor, the mounting position must be self-draining, avoid horizontal surfaces and pooling of liquids, be easily accessible for maintenance and inspection, and facilitate CIP cleaning.
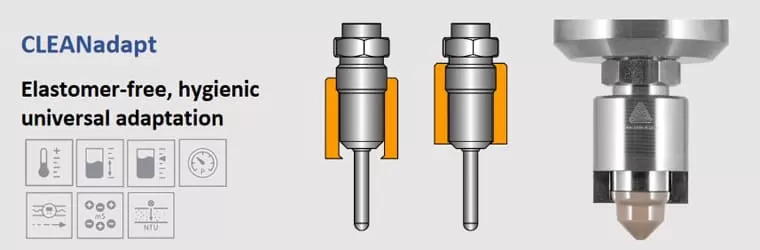
With appropriate system planning, this can be achieved with all common process connections. However, due to its extensive experience in this field, Anderson-Negele has also developed special process connection systems with a wide range of different Process Adapters and Process Connections for food, beverage and biopharmaceutical equipment based on the guiding principle “Hygienic By Design”.
Besides the systems CLEANadapt and FLEXadapt for the Food & Beverage industry we also offer process connections that were specially developed for the superior requirements of pharmaceutical and life-science applications: PHARMadapt ESP, PHARMadapt EPA and CPM.

.webp&w=3840&q=75)


